Hepatitis in a Febrile Infant with Coronavirus Disease 2019 (COVID-19)
AUTHORS:
Juan Cardenas Fimbres, MD*; Jessica Warrick-Imrisek, MD; Nizar F Maraqa, MD, FPIDS
Department of Pediatrics, University of Florida College of Medicine Jacksonville
*Corresponding Author
Case Report | PUBLISHED FALL 2022 | Volume 42, Issue 4
DOWNLOAD PDF
Abstract
We report a 7-week-old infant who presented with fever and transaminitis as a manifestation of infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The infant was evaluated for common age-related etiologies of hepatitis, ruling those out. As the coronavirus disease 2019 (COVID-19) pandemic is expected to cause more cases in the unvaccinated pediatric population, we emphasize the importance of considering SARS-CoV-2 infection in the differential diagnosis of hepatitis in infants under 1 year of age, in addition to the other age-related etiologies.
Introduction
While fever and respiratory symptoms have been the most common manifestations of SARS-CoV-2 infection in young children, less common manifestations with varied prognostic implications have been identified. 1, 2 Specifically, liver involvement has been described in COVID-19 and correlated with mortality in adult studies.3 Little information is available about hepatitis in young children with COVID-19.
We report a 7-week-old infant with fever and transaminitis where more common etiologies of hepatitis were ruled out, leaving SARS-CoV-2 as the most likely cause. SARS-CoV-2 hepatitis can mimic other frequent etiologies of hepatitis in febrile infants and should be considered in the differential diagnosis.
Clinical Course
A 7-week-old male infant presented to the emergency department (ED) with fever (a rectal temperature of 38.3°C), cough, nasal congestion, and a decrease in oral intake. He was born at full term via vaginal delivery with no significant prenatal maternal history and no perinatal or postnatal complications. His father and mother tested positive by polymerase chain reaction (PCR) for SARS-CoV-2 two days prior to his presentation. The mother developed fever and breathing difficulty for which she was hospitalized while the father remained asymptomatic.
On initial physical examination in the ED, the infant appeared inactive, but well hydrated and not fussy. He had a rectal temperature of 39°C, heart rate (HR) of 147 beats/minute, respiratory rate of 34 breaths/minute, blood pressure of 124/62 mmHg, and SpO2 of 100% on room air. A blood culture, urinalysis and culture, complete blood count (CBC), comprehensive metabolic panel (CMP), cerebrospinal fluid (CSF) analysis and culture, as well as an upper respiratory viral PCR panel (including SARS-CoV-2) were collected. A dose of ceftriaxone was administered intravenously, and the patient was admitted to the general floor for further management.
The initial workup was remarkable for a bloody CSF that was uninterpretable for cell analysis, with moderate turbidity, protein of 138 mg/dL (reference range: 12-60), and glucose of 54 mg/dL (reference range: 45-75). He had a peripheral white blood cell (WBC) count of 5.4 x103/µL (reference range: 5-19.5), 78% lymphocytes, 17% neutrophils and 2% band forms with an absolute neutrophilic count (ANC) of 0.7 x103/µL (reference range: 1.5-10). His upper respiratory viral PCR panel was positive for SARS-CoV-2 as well as Rhinovirus/Enterovirus. He had a serum aspartate aminotransferase (AST) of 121 IU/L (reference range: 10-60), alanine aminotransferase (ALT) of 54 IU/L (reference range: 17-63, alkaline phosphatase of 357 IU/L (reference range: 67-391), total bilirubin of 1.4 mg/dL (reference range: 0.3-1.8), protein of 5.7 g/dL (reference range: 4.4-6.8), and albumin of 3.8 g/dL (reference range: 2.7-4.7).
On hospital day 2 (HD#2), he remained febrile (maximum rectal temperature of 38.4°C) and on room air. Repeat testing revealed an increasing AST to 323 IU/L and ALT to 123 IU/L with worsening neutropenia (ANC of 0.40 x103/µL). On HD#3, his fever continued with a further increase of his liver transaminases (AST 468, ALT 468). Ceftriaxone was discontinued after blood, urine, and CSF cultures remained without bacterial growth at 48 hours. An ultrasound of the abdomen demonstrated an enlarged echogenic liver with no focal lesions.
The fever resolved on HD#5. The ANC remained low at 0.70 x103/µL but the liver transaminases continued rising. Ferritin level peaked at 9,284 µg/L (reference range: 24-336) as shown in Table 1. His C-reactive protein (CRP) was 0.1 mg/dL (reference range: 0.5-1), B-natriuretic peptide (BNP) was 22 pg/mL (reference range: 1-100), and prothrombin time was 12.7 seconds (reference range: 11.8-15). Additional workup recommended by the Infectious Diseases consulting service included SARS-CoV-2 IgG, blood PCR for cytomegalovirus (CMV), herpes simplex virus (HSV), and enterovirus as well as hepatitis C antibody and serum lactate dehydrogenase (LDH). The infant’s physical examination continued to be unremarkable, and he was feeding both breast milk and formula appropriately.
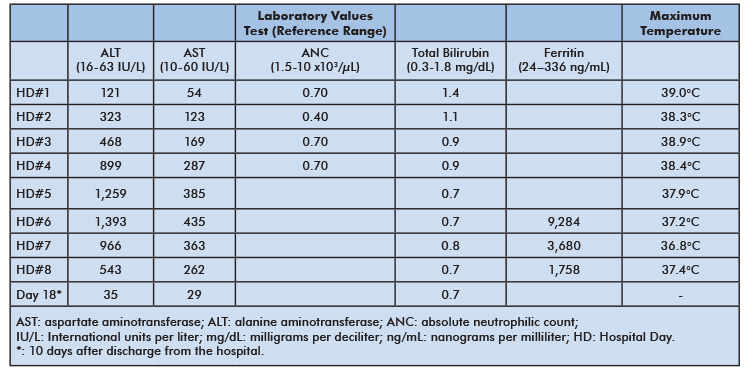
Table 1: Select laboratory values and corresponding maximum daily temperature
After peaking on HD#6, the serum transaminases and ferritin level gradually decreased until the patient was discharged on HD#8. Workup for the other potential etiologies for hepatitis came back negative. Ten days following discharge, his AST, ALT, and LDH levels were back within normal limits.
Discussion
The majortiy of children infected with SARS-CoV-2 are asymptomatic or have mild symptoms of COVID-19. Compared to adults, children with COVID-19 tend to have a better outcome.2 In a metanalysis that included 5,829 pediatric patients who tested positive for SARS-CoV-2, Cui et. al. reported that 20% were asymptomatic, 33% had mild symptoms (described as an acute upper respiratory infection or isolated digestive symptoms), 51% had a moderate illness (i.e. pneumonia without hypoxia or difficulty breathing), 7% had severe pneumonia with hypoxia and dyspnea and 5% were critical (presenting with or rapidly progressing to acute respiratory failure). The authors noted that there was a higher proportion of critically ill cases among infants < 1 year of age compared to older children.2
Most symptomatic children with COVID-19 present with respiratory symptoms. Commonly described manifestations include fever, cough, sore throat, nasal congestion, and rhinorrhea.1 Less frequently, hepatic, pancreatic, cardiac, renal, and lymphocyte alterations have been described in acute COVID-19.1-4 Additionally, patients with COVID-19 who have no pre-existing liver disease have been described to have abnormal levels of AST and ALT.4, 5 However, in the majority of these cases, the liver involvement is usually mild and self-limited.5
In the febrile infant we present here, the liver enzymes peaked as the fever resolved and respiratory symptoms subsided. While a liver biopsy was not performed for a definitive etiologic diagnosis due to the favorable course and the invasive nature of the procedure, other potential causes of infectious hepatitis were investigated. This additional workup detected no viremia for enterovirus, CMV, or HSV, a negative hepatitis C serology, and negative maternal hepatitis B surface antigen status which make these unlikely etiologies of the infant’s hepatitis.
SARS-CoV-2 has been described to cause direct damage to liver parenchyma in COVID-19 patients who had a liver biopsy, suggesting tropism of the virus for the hepatocytes.4 SARS-CoV-2 transmission has been reported after allograft transplantation of liver parenchyma.6
It remains inconclusive if SARS-CoV-2 can be transmitted vertically, but cases of neonates that test positive for SARS-CoV-2 within the first 24 hours of life without significant exposure, other than at the time of delivery, have been reported.6, 7
Conclusions
As the healthcare community continues to deal with the COVID-19 pandemic and the impacts of emerging new variants with greater infectivity,8 especially on the unvaccinated pediatric population, we present this infant’s case to alert pediatricians to the possibility of hepatitis due to SARS-CoV-2 among the very young. SARS-CoV-2 should be part of the differential diagnosis of acute hepatitis in febrile infants. Additionally, while elevated ALT levels have been directly correlated with mortality in adults with COVID-19, this has not been observed in infants and young children.3 Our infant’s hepatitis resolved spontaneously as he recovered from COVID-19, but it may be important for pediatric providers to follow the liver enzyme levels to ensure their return to normal. More data need to be collected to better describe the liver involvement in infants who develop COVID-19.
References
- Cui, X, Zhao, Z, Zhang, T, et al. A systematic review and meta-analysis of children with coronavirus disease 2019 (COVID-19). J Med Virol 2021;93(2):1057-1069.
- Ludvigsson, JF. Systematic review of COVID-19 in children shows milder cases and a better prognosis than adults. Acta Paediatr 2020;109(6):1088-1095.
- Papadopoulos, N, Vasileiadi, S, Deutsch, M. COVID-19 and liver injury: where do we stand? Ann Gastroenterol 2020;33(5):459-464.
- Sun, J, Aghemo, A, Forner, A, et al. COVID-19 and liver disease. Liver Int 2020;40(6):1278-1281.
- Su, TH, Kao, JH. The clinical manifestations and management of COVID-19-related liver injury. J Formos Med Assoc 2020;119(6):1016-1018.
- Alzamora, MC, Paredes, T, Caceres, D, et al. Severe COVID-19 during Pregnancy and Possible Vertical Transmission. Am J Perinatol 2020;37(8):861-865.
- De Rose, DU, Piersigilli, F, Ronchetti, MP, et al. Novel Coronavirus disease (COVID-19) in newborns and infants: what we know so far. Ital J Pediatr 2020;46(1):56.
- Centers for Disease Control and Prevention. National Center for Immunization and Respiratory Diseases (NCIRD). Division of Viral Diseases. Delta Variant: What We Know About the Science; 2021. https://www.cdc.gov/coronavirus/2019-ncov/variants/delta-variant.html. Accessed October 21, 2021.
