Glucagon-like Peptide 1 Receptor Agonists in the Treatment of Pediatric Obesity
AUTHORS:
Kalie Nuss, BS1; Patricia Emmanuel, MD2
1University of South Florida Morsani College of Medicine
2USF Health Department of Pediatrics, Division of Infectious Disease
REVIEW ARTICLE | PUBLISHED Spring 2026 | Volume 46, Issue 2
DOWNLOAD PDF
Abstract
Pediatric obesity is increasing globally. While numerous lifestyle interventions, pharmacologic therapies, and surgical options are widely available for adults, these options are limited in pediatric patients. Obesity in this population is especially detrimental as it can impact the child’s social and physical health as well as put them at risk for the development of several other comorbidities in adulthood. Within the last decade, novel therapies, including glucagon-like peptide 1 receptor agonists, have been studied for their use as adjuvants to weight loss therapy. Over the last few years, many of these medications have been approved for widespread use in adults with obesity and have had heralding success. As such, glucagon-like peptide 1 receptor agonists are being more carefully studied for use in pediatric patients with obesity. Several trials have been completed, and as of this review, two glucagon-like peptide 1 receptor agonists have been approved for some cases in this population. Many trials are still in progress, testing the utility and safety of these medications in younger children. However, there is still concern regarding the effects of these medications on the child’s growth and development and their proposed long-term consequences.
Introduction
In 1997, pediatric obesity was declared an epidemic by the World Health Organization, and currently, in the United States, 1 in 5 children and adolescents meet the definition of obesity.1 Obesity is defined as having a body mass index (BMI) at or above the 95th percentile for age and sex. Unfortunately, this number is still on the rise, and it has been predicted that one-third of children and adolescents will be overweight or have obesity by 2050.2 Many factors contribute to this trend including, social and economic determinants such as, access to healthy foods, food deserts, access to safe spaces for recreation, excessive screentime, bullying, and the child’s perception of food security. The recent pandemic has also contributed to social isolation and the popularization of the sedentary lifestyle. Other factors that influence childhood obesity include the weight of the child’s parents, race and ethnicity, and the child’s birth weight.3 There are many other genetic and environmental factors that may also contribute to childhood obesity.
Adverse Effects and Long-Term Consequences of Pediatric Obesity
It is well known that obesity in adults causes a cascade of metabolic derangements, including type 2 diabetes (T2DM), chronic inflammation, and cardiovascular disease. Similar to adults, children with obesity have a higher chance of being diagnosed with T2DM and hypertension during childhood. One study shows that as high as 37% of hypertension (HTN) in children is associated with childhood obesity.4 Additionally, there is a higher prevalence of non-alcoholic fatty liver disease in children with obesity, a higher incidence of asthma, and a higher fracture rate in these patients.4 It appears that obesity also influences puberty, especially in females, due to higher levels of adrenal androgen production that are converted to estrogens. Furthermore, higher levels of leptin in children with obesity may lead to CNS stimulation that also impacts the onset of puberty.5 It has also been demonstrated that children with obesity live with a chronic low level of inflammation, which has been detected in children as young as preschool age. This inflammation, as well as other changes caused by obesity, leads to a worse quality of life in the future and has been linked to higher levels of insulin resistance in adulthood. Obesity and chronic inflammation have also been linked with a higher chance of developing rheumatoid arthritis and inflammatory bowel disease. Children with obesity have a higher risk for issues in adulthood, including osteoarthritis, multiple sclerosis, T2DM, and several cancers.4 The complex nature of this disease and its long-term consequences warrant a multifactorial approach to treatment.
Currently Available Therapies and Guidelines for Managing Pediatric Obesity
The American Academy of Pediatrics (AAP) has carefully evaluated various therapies for managing pediatric obesity and has subsequently designed specific guidelines to help providers decide optimal treatment plans.6 The suggested therapies and their supporting evidence have been listed in Table 1. The United States Preventive Services Task Force (USPSTF) currently recommends that children and adolescents over the age of 6 years should receive comprehensive, intensive behavioral interventions such as interventional health behavior and lifestyle treatment (IHBLT) as described by the AAP.7 However, this therapy is time-intensive, requiring frequent visits, involves the other members of the family unit, and can be difficult to coordinate. Additionally, IHBLT is not widely offered, and few primary care physicians receive adequate training to administer such therapy. Because IHBLT may be challenging to implement, other forms of therapy, such as bariatric surgery and pharmacotherapy, are available to assist with weight loss, especially for patients who do not respond to IHBLT. Bariatric surgery may be available to pediatric patients with a BMI of >35 Kg/m2 and a comorbidity such as T2DM, polycystic ovary syndrome, or patients whose BMI exceeds 40 Kg/m2.6 Usually limited to age 13 years and above, bariatric surgery has demonstrated success but still has limited uptake (Table 1). Pharmacotherapy is typically used as an adjunct treatment, and the evidence supporting its use varies among patients. As it stands, no medication is included in the USPSTF recommendation, and more research is needed into promising new therapies, such as glucagon-like peptide-1 receptor agonists (GLP-1RA), which will be the focus of this review.
GLP-1RAs have gained popularity in treating obesity in adults, and there is sufficient evidence supporting their use for weight loss. Because of their recent popularity, these drugs are also being heavily considered and currently studied for their use in treating pediatric obesity as well. This review will discuss GLP-1RAs and their current and future use in pediatric patients with obesity, and it will address barriers to and concerns regarding their use as a therapy for weight loss in this population.
Rationale for Glucagon-like Peptide 1 Receptor Agonist Use in Obesity Management
The human body constantly measures and assesses its blood glucose levels and activates various pathways to maintain homeostasis. After a meal, when blood glucose levels increase, insulin is released and sets off a cascade of events to help the body properly store glucose and keep it from releasing added endogenous glucose into the bloodstream. The converse happens through the actions of glucagon, cortisol, and growth hormone, which promote endogenous glucose release during periods of starvation.
There are many other molecules involved downstream of these pathways, and even more that help initiate these pathways. One such molecule, glucagon-like peptide 1 (GLP-1), is released from L-cells in the intestinal tract and plays a role in stimulating insulin secretion. The most well-understood mechanism for the release of GLP-1 involves nutrients such as lipids and carbohydrates coming into contact with microvilli in the intestinal tract. However, there may also be neurohormonal mechanisms for its release involving stimulation from the vagus nerve that are less understood.12 Once GLP-1 is released, it acts on the G-protein coupled receptor, GLP-1 receptor (GLP-1R), which is located on a variety of cell types, including pancreatic beta cells and cells of the brain, heart, gastrointestinal tract, and kidneys. Due to the widespread distribution of GLP-1R, GLP-1 has a multitude of effects throughout the body, though its primary functions include reducing gastric motility and amplifying meal-associated insulin secretion while reducing glucagon secretion. As such, targeting GLP-1R has become a prominent method for increasing insulin secretion in the treatment of T2DM, and it has been recently approved in adults as a therapy for obesity due to its ability to slow gastric emptying. In the body, GLP-1 is rapidly degraded by dipeptidyl peptidase IV (DPP-4), another target used in the management of T2DM. Because of its rapid degradation and removal from the body, giving GLP-1 as a therapy was unsuccessful. The molecule was modified to be resistant to DPP-4 and to decrease its renal clearance, while retaining the same blood glucose-lowering properties. This novel therapy was first approved for the treatment of T2DM in 2005. In 2014, the first GLP-1RA, liraglutide, was approved for weight loss in adults, even without a diagnosis of T2DM.13 Since then, several other GLP-1RAs have undergone clinical trials and have been approved for this use in adults, and they have become increasingly readily available to the public over the last few years.
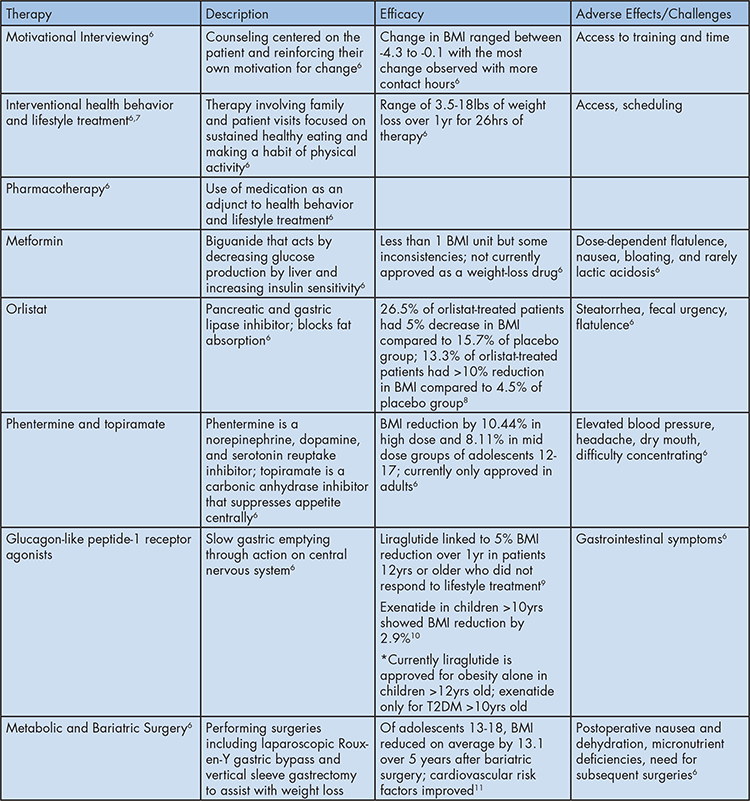
Table 1: AAP Recommended Therapies for Pediatric Obesity
Legend: This table provides a list of AAP-recommended therapies to use when considering treatment options for pediatric obesity. A brief description of each therapy, in addition to evidence supporting its use and any adverse effects noted in the literature, has been included to provide a broader understanding of available treatment options.
Current State of GLP-1RA Use in Pediatric Medicine
Because of the widespread success of GLP-1RAs in adults and in light of treatment challenges, there has been significant motivation to learn more about how they may benefit the pediatric population. As of the time of this review, several GLP1-RAs, including dulaglutide, liraglutide, exenatide, and semaglutide, have been approved to be used in the treatment of T2DM in children older than 10 years when insulin and metformin do not provide sufficient glycemic control. Nonetheless, little is known about the effects of their long-term use in children younger than 10 years, therefore their use in young children is not recommended. More research is needed to determine safety and efficacy.
Recently, two of these drugs were also approved to manage obesity in children 12 years and older. Novel research regarding these compounds focuses on confirming their safety and efficacy in this population and seeking approval for use in younger children as well. There are also several ongoing clinical trials assessing the long-term effects of these medications, especially in pediatric patients. Due to the newness of these compounds and a lack of sufficient research on possible side effects in pediatric patients, several concerns remain regarding their broad-scale use as a weight loss therapy.
Approved Use of GLP-1RAs in Pediatric Patients with Obesity
Until lately, there were very few drugs approved by the Food and Drug Administration (FDA) for weight loss in pediatric patients. However, in recent years, several GLP-1RAs have been shown in clinical trials to increase weight loss in pediatric patients as an adjunct therapy when lifestyle modifications alone have proven insufficient. As such, two GLP-1RAs have been FDA-approved for weight loss in pediatric patients aged 12 years and older due to their demonstrated benefits and limited side effects.
One of the currently approved GLP-1RAs is liraglutide, which has been shown to reduce BMI when added to lifestyle modifications in adolescents over the age of 12 years. In one randomized, double-blind trial involving 251 adolescents aged 12-18 years with obesity who did not respond well to lifestyle modification alone, adding liraglutide was shown to increase weight loss.9 Liraglutide therapy featuring a 3.0 mg dose subcutaneously once a day in addition to lifestyle therapy was compared to a placebo treatment in patients also adhering to the same lifestyle modifications. Participants received therapy for 56 weeks, followed by a 26-week follow-up period. In this trial, liraglutide yielded a significantly greater reduction in BMI, with an average loss of 4.64 percentage points in the treatment group compared to placebo. In addition, more participants in the treatment group experienced a 5% reduction (43.3% treatment vs 18.7% in placebo) and a 10% reduction (26.1% treatment vs 8.1% placebo) in BMI. Participants receiving liraglutide also lost, on average, 4.5 kg more weight than those in the placebo group. Few participants in either group noted serious adverse events, but gastrointestinal side effects were more common in the treatment group.9
The only other GLP-1RA that is currently FDA-approved to be added to lifestyle therapy in the treatment of pediatric obesity in adolescents between the ages of 12 and 17 years is semaglutide. This drug was approved after liraglutide, and its efficacy was demonstrated in several clinical trials. In one double-blind, parallel-group, randomized, placebo-controlled trial, semaglutide was assessed as a weight-loss therapy in children 12-18 years of age with obesity (BMI in the 95th percentile or higher) or who were overweight (BMI in the 85th percentile or higher) but who had at least one comorbid condition related to their weight.14 Participants either received a 2.4 mg subcutaneous dose of semaglutide once a week or a once-weekly placebo in addition to lifestyle interventions for 68 weeks. Two hundred and one participants were enrolled, and 180 completed the study. This study demonstrated that the addition of semaglutide to lifestyle therapy produced a greater decrease in BMI than lifestyle modification alone. Participants receiving semaglutide experienced a BMI reduction of 16.0 percentage points compared to those in the placebo group, where the BMI increased by 0.6. Furthermore, 95 out of 131 (73%) participants in the treatment group achieved 5% or more weight loss compared to only 11 out of 62 (18%) participants in the placebo group. Additionally, improvement of cardiometabolic risk factors, including glycated hemoglobin, lipids, alanine aminotransferase levels, and waist circumference, was noted in the semaglutide group compared to placebo. There was a greater incidence of gastrointestinal adverse events (62% vs 40% placebo) and cholelithiasis (4% compared to 0% of placebo) in the group that received semaglutide. Only a small percentage of participants experienced serious adverse events in either group (11% in the treatment group, 9% in the placebo group).14 While there are several ongoing clinical trials assessing the use of semaglutide in younger children, there has been some evidence of its success in reducing BMI in children as young as 10 years. One retrospective observational study investigated the effect of semaglutide on the BMI of children aged 10-18 years in the UK who were receiving treatment in addition to lifestyle management.15 The treatment produced a reduction in BMI standard deviation scores at 6 and 12 months of 0.32±0.27 and 0.54±0.52, respectively. Patients also achieved an average of 7.03±7.50 kg of weight loss. The most common side effects were mild gastrointestinal issues, and of the 50 patients studied, one patient developed gallstones.15 Though semaglutide appears to be safe and efficacious in children as young as 10 years, it remains unapproved, as more research is needed. Despite this, it has been occasionally prescribed off-label in younger children on a case-by-case basis.
Future Directions and Update on Current Clinical Trials Featuring GLP-1RA Use in Pediatric Patients with Obesity
Though currently only two GLP-1RAs have been officially FDA-approved for the purpose of aiding in weight loss in adolescents, several others have been undergoing clinical trials. Such trials include investigating other GLP-1RAs’ use in adolescents, the use of GLP-1RAs in young children, and trials involving various possible adverse effects of using these medications in pediatric patients.
As discussed, liraglutide is approved for adolescents aged 12-18 years as an adjunct to lifestyle modification for children who have had an insufficient response to lifestyle therapy alone. The SCALE Kids trial, published in 2024, aimed to assess the efficacy of liraglutide in children aged 6 to less than 12 years as an alternative therapy for children of this age group who are unable to achieve weight loss with traditional treatments.16 This study is similar to the trials conducted in adolescents, featuring the same 3.0 mg daily subcutaneous dosage in addition to the lifestyle modifications compared with placebo for 56 weeks, followed by a 26-week follow-up period. It featured 82 participants randomized to receive either a 3.0 mg dose of liraglutide or a placebo and demonstrated that among children in this age range with obesity, treatment with liraglutide resulted in a greater BMI reduction than lifestyle modification alone. After 56 weeks, the mean changes in BMI for the treatment and placebo groups were -5.8% and +1.6%, respectively. One difference between the results of the liraglutide study in this age group and in children 12-18 years was the rate of adverse effects.16 In this trial, 80% of participants receiving liraglutide endorsed gastrointestinal adverse events, whereas in the previously mentioned trial, only 64.8% of participants receiving liraglutide had these symptoms.9,16 Additionally, this trial reported 12% of participants receiving treatment experienced serious adverse events, while only 2.4% of children ages 12-18 years in the other study had these effects.9,16 Though this trial suggests that liraglutide is effective in children ages 6-12 years, more research is required regarding long-term effects and to confirm liraglutide’s efficacy in this age group before it could be approved for use by the FDA. Semaglutide, which is also approved for adolescents ages 12-18 years, will also be studied as a weight management therapy in children as young as age 6 years. Though this study, STEP Young, has not yet begun recruitment, it plans to follow children and teenagers for 132 weeks as they receive once weekly semaglutide to assess the medication’s effect on BMI.17
Another GLP-1RA, tirzepatide, that has had recent success in adults, is currently undergoing several trials for use in pediatric patients. This drug is a dual GLP-1RA and glucose-dependent insulinotropic peptide (GIP) agonist, which means it also functions like GIP to increase insulin secretion and slow gastric emptying. One trial aimed to investigate the efficacy, safety, and pharmacokinetics of tirzepatide in pediatric and adolescent patients in the age range of 6-11 years with obesity. This study began in 2023 and is set to be completed by January 2025, though the results have not yet been released.18 Another trial featuring tirzepatide, SURMOUNT-ADOLESCENTS-2, is currently in phase III. This trial enrolled pediatric patients between 12 and 17 years of age with obesity and at least 2 weight-related comorbidities such as HTN, prediabetes, or hypertriglyceridemia. In addition, participants must have had at least one unsuccessful dietary attempt at weight loss. The primary outcome of this study will be the percent change from BMI at week 72 compared to baseline, and several secondary outcomes related to BMI, including waist circumference, and other weight-related conditions. This trial is estimated to be completed by the end of May in 2027.19
Exenatide is yet another relevant GLP-1RA in this discussion, as it has been approved for use in children aged 10-18 years for the treatment of T2DM, but it has not yet been approved as a weight loss therapy. One randomized, double-blind, parallel, placebo-controlled clinical trial conducted in 2020 investigated the effects of extended-release (ER) exenatide primarily on the BMI of children in that age group with a BMI above 30 Kg/m2. Forty-four participants were selected to receive 2 mg ER exenatide or placebo once weekly for 6 months along with lifestyle therapy. At the end of the trial, it was determined that treatment with exenatide led to a moderate reduction in BMI and improvement in glucose tolerance and cholesterol. The study additionally reported that the medication was well-tolerated.10 Another trial was conducted in 2022 to assess ER exenatide at a dose of 2.0 mg as an adjunct to meal replacement therapy and also focused on children 12-18 years of age with obesity. The study followed 100 participants who had been enrolled in meal replacement therapy and experienced BMI reduction before the addition of weekly exenatide or placebo to determine if exenatide would influence BMI rebound in these adolescents. The results of the study, however, were not statistically significant, but they do in part suggest that exenatide could help reduce BMI rebound in adolescents who had lost weight initially with dietary interventions.20
Concerns about the Use of GLP-1RAs in Pediatric Patients
In the current literature, the most commonly reported adverse effects associated with the use of GLP-1RAs in pediatric patients are gastrointestinal, including vomiting, diarrhea, abdominal pain, or hypoglycemia. More severe side effects include gallstones and pancreatitis, and they are contraindicated in patients at risk for medullary thyroid cancer. As the number of adults using GLP-1RAs expands, rare side effects have been reported, such as severe gastroparesis and non-arteritic ischemic optic neuropathy.21 Since their recent increase in popularity and approval for use in some pediatric patients, more concerns have arisen regarding their effects on growth and development, mental health, and possible long-term consequences.
Pediatric patients differ from adult patients in many ways, one being energy expenditure. In children, additional energy is needed for growth and development. There is a delicate balance of energy intake and expenditure required in this population for sufficient growth and development to occur. One concern of using these medications, which inherently reduce energy intake through various satiety mechanisms, is that this delicate balance could be upset.22 It has been postulated that this may result in defective growth and musculoskeletal development. There is currently a clinical trial to determine how GLP-1RAs may alter bone density, structure, and strength in adolescents. This trial will follow children and young adults aged 12-21years who are taking semaglutide for two years to assess the impact of GLP-1RAs on skeletal integrity.23
Another area of concern being discussed in the current literature addresses the abuse potential of these medications and their effects on the mental health of adolescent patients. Because of the widespread nature of their effectiveness portrayed in the media, GLP-1RAs have a reputation for inducing quick and effective weight loss. As such, it is likely that older adolescents at least have heard of these medications and know about their use in weight loss. Adolescents are generally a vulnerable population when it comes to body image and self-perception, and for them, these drugs provide another possible method of quickly achieving their ideal weight status, resulting in a high potential for abuse. In a similar fashion, it has been proposed that these medications may be misused by student athletes and other highly competitive students who must meet specific weight requirements for competitions or to enhance athletic performance.22 Though these medications are not widely prescribed to the pediatric population currently, there has been a recent trend of increasing medicalization of pediatric conditions.22 As such, if these drugs gain approval, it logically follows that pediatric obesity could become one of these conditions, leading to widespread use and availability of these medications to an already vulnerable population.
As GLP-1RAs have gained popularity, some cases involving rarer adverse effects have been reported in recent literature. One case report from 2024 involved an obese adolescent on low-dose liraglutide who presented with vomiting, epigastric pain, and decreased urine output and was diagnosed with an acute kidney injury (AKI) in addition to hepatic dysfunction. It was determined that liraglutide was the cause of his presentation, and the medication was discontinued.24 A study conducted in 2022 demonstrated a correlation between GLP-1RAs and AKI in adult patients. Of the cases of AKI reported, liraglutide was the most common GLP-1RA culprit, most commonly occurring in patients between the ages of 45 and 84 years. AKI is a possible serious adverse effect associated with GLP-1RA use and should be considered when deciding whether to prescribe this medication.25
Conclusions
Pediatric obesity is a serious condition that affects a significant portion of today’s children and young adults, and it continues to rise in prevalence. Because this condition has detrimental long-term consequences that threaten the child’s quality of life in adulthood, it is incumbent that new therapies be made available to help treat obesity in this population. It is a complex problem that requires a multifaceted approach. GLP-1RAs have become targets for new research into possible remedies for the pediatric obesity epidemic. Due to their success in reducing BMI in adults who have struggled to lose weight with lifestyle modification alone.
Several trials have been conducted that demonstrate the promise of these medications in younger children, and still, more are set to be completed within the next few years, with the hope of gaining approval for additional GLP-1RAs. Today, two GLP-1RAs, liraglutide and semaglutide, are approved for the purpose of weight loss as an adjunct to lifestyle therapy. They are only approved for children over the age of 12 years. A recent MMWR study of national prescribing data demonstrated that less than 1% of obese adolescents have been prescribed GLP-1RAs.1
Even with FDA approval, there is limited insurance coverage for these agents, and Medicaid, which covers about 40% of children in the United States, does not cover this indication26, at least in the state of Florida. Without insurance, patients may be required to pay $1000 per month out of pocket for these medications. This cost barrier creates a socioeconomic disparity for patients who cannot afford private insurance or the high costs of these medications. Aside from high costs, other reasons for such a low rate of GLP-1RA prescription include production shortages and concerns for possible adverse effects.1
Despite their demonstrated success in this population, there are still many concerns regarding long-term effects, and no data on outcomes are available concerning long-term use. While the main side effects noted in the trial are mild to moderate gastrointestinal effects, other serious events have been reported in the literature. There are clinical trials that are currently studying long-term complications. Because of the paucity of completed research and data regarding their performance and tolerance in this population in the long-term, it is essential to weigh the individual benefits and risks of using these medications on a case-by-case basis and to monitor long-term outcomes carefully.
Acknowledgments: We would like to acknowledge and thank Dr. Dorothy Shulman for her help and contribution to this paper.
Author Disclosure: The authors report no conflicts of interest.
References
- Kompaniyets L, Pierce SL, Porter R, et al. Prescriptions for obesity medications among adolescents aged 12-17 years with obesity—United States, 2018-2023. MMWR. 2025;74 (20):337-344.
- GBD 2021 Adolescent BMI Collaborators. Global, regional, and national prevalence of child and adolescent overweight and obesity, 1990-2021, with forecasts to 2050: A forecasting study for the Global Burden of Disease Study 2021. Lancet. 2025;405 (10481):785-812.
- Jebeile H, Kelly AS, O’Malley G, Baur LA. Obesity in children and adolescents: Epidemiology, causes, assessment, and management. Lancet Diabetes Endocrinol. 2022;10 (5):351-365.
- Marcus C, Danielsson P, Hagman E. Pediatric obesity—long-term consequences and effect of weight loss. J Intern Med. 2022;292 (6):870-891.
- Huang A, Reinehr T, Roth CL. Connections between obesity and puberty. Curr Opin Endocr Metab Res. 2021;14:160-168.
- Skinner AC, Staiano AE, Armstrong SC, et al. Appraisal of clinical care practices for child obesity treatment. Part I: interventions. Pediatrics. 2023;151 (2):e2022060642.
- U.S. Preventive Services Task Force. High body mass index in children and adolescents: Interventions. USPSTF Recommendations. Published June 18, 2024. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/obesity-in-children-and-adolescents-screening. Accessed June 26, 2025.
- Chanoine JP, Hampl S, Jensen C. Effect of orlistat on weight and body composition in obese adolescents. JAMA. 2005;298 (23):2873-2883.
- Kelly AS, Auerbach P, Barrientos-Pérez M, et al. A randomized, controlled trial of liraglutide for adolescents with obesity. N Engl J Med. 2020;382 (22):2117-2128.
- Kelly AS, Metzig AM, Rudser KD, et al. Exenatide as a weight-loss therapy in extreme pediatric obesity: A randomized, controlled pilot study. Obesity. 2012;20 (2):364-70.
- Olbers T, Beamish AJ, Gronowitz E, et al. Laparoscopic Roux-en-Y gastric bypass in adolescents with severe obesity (AMOS): A prospective, 5-year, Swedish nationwide study. Lancet Diabetes Endocrinol. 2017;5 (3):174-183.
- Holst JJ. The physiology of glucagon-like peptide 1. Physiol Rev. 2007;87 (4):1409-1439.
- Crane J, McGowan B. The GLP-1 agonist, liraglutide, as a pharmacotherapy for obesity. Ther Adv Chronic Dis. 2015;7 (2):92-107.
- Weghuber D, Barrett T, Barrientos-Pérez M, et al. Once-weekly semaglutide in adolescents with obesity. N Engl J Med. 2022;387 (24):2245-2257.
- van Boxel EJ, Rahman S, Lai K, Boulos N, Davis N. Semaglutide treatment for children with obesity: An observational study. Arch Dis Child. 2024;109 (10):822-825.
- Fox CK, Barrientos-Pérez M, Bomberg EM, et al. Liraglutide for children 6 to 12 years of age with obesity—A randomized trial. N Engl J Med. 2025;392 (6):555-565.
- A research study on how well semaglutide helps children and teenagers with excess body weight lose weight (STEP Young). Clinicaltrials.gov identifier: NCT05726227. Updated January 30, 2025. https://clinicaltrials.gov/study/NCT05726227. Accessed June 26, 2025.
- A study of tirzepatide (LY3298176) in pediatric participants with obesity. Clinicaltrials.gov identifier: NCT05696847. Updated February 4, 2025. https://clinicaltrials.gov/study/NCT05696847. Accessed June 26, 2025.
- A study of tirzepatide in adolescents with obesity and weight-related comorbidities (SURMOUNT-ADOLESCENTS-2). Clinicaltrials.gov identifier: NCT06439277. Updated June 25, 2025. https://clinicaltrials.gov/study/NCT06439277. Accessed June 26, 2025.
- Fox CK, Clark JM, Rudser KD, et al. Exenatide for weight-loss maintenance in adolescents with severe obesity: A randomized, placebo-controlled trial. Obesity. 2022;30 (5):1105-1115.
- Hsu AY, Kuo H, Wang Y, et al, Semaglutide and nonarteritic anterior ischemic optic neuropathy risk among patients with diabetes. JAMA Ophthalmol. 2025;143 (5):400-407.
- Cooper DM, Rothstein MA, Amin A, Hirsch JD, Cooper E. Unintended consequences of glucagon-like peptide-1 receptor agonists medications in children and adolescents: A call to action. J Clin Transl Sci. 2023;7 (1):e184.
- Bone metabolism in 12-21 year olds undergoing GLP-1 receptor agonist therapy. Clinicaltrials.gov identifier: NCT06903923. Updated April 4, 2025. https://clinicaltrials.gov/study/NCT06903923. Accessed June 26, 2025.
- Komagodski R, Wittenberg A, Bahat H, Rachmiel M. Acute kidney and liver injury associated with low-dose liraglutide in an obese adolescent patient. Pediatrics. 2024;154 (1):e2023063719.
- Dong S, Sun C. Can glucagon-like peptide-1 receptor agonists cause acute kidney injury? An analytical study based on post-marketing approval pharmacovigilance data. Front Endocrinol. 2022;13:1032199.
- Butterfield R. The advent of GLP-1 use for pediatric obesity. AAP Journal Blogs. Published February 13, 2025. https://publications.aap.org/journal-blogs/blog/31278/The-Advent-of-GLP-1-Use-for-Pediatric-Obesity. Accessed June 26, 2025.
